Momsy Nasal Spray Ensures Free & Effortless Nasal Breathing for Hours
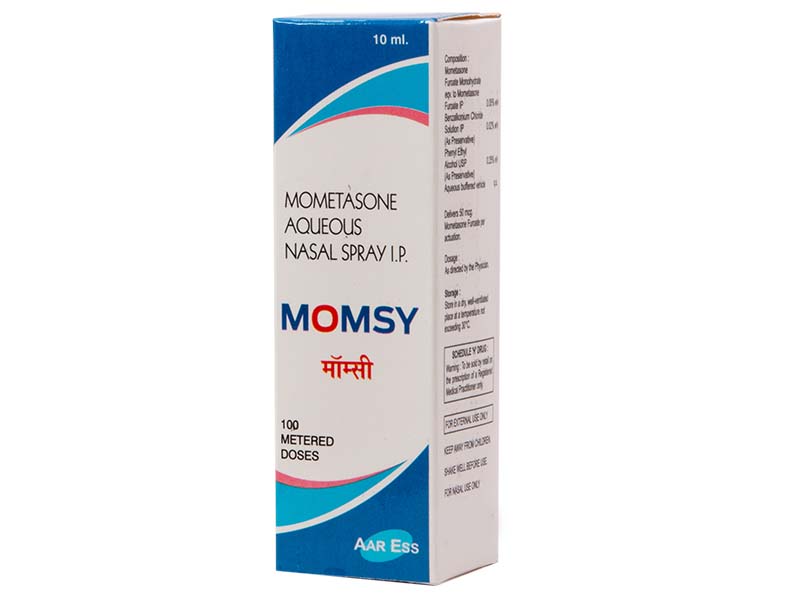

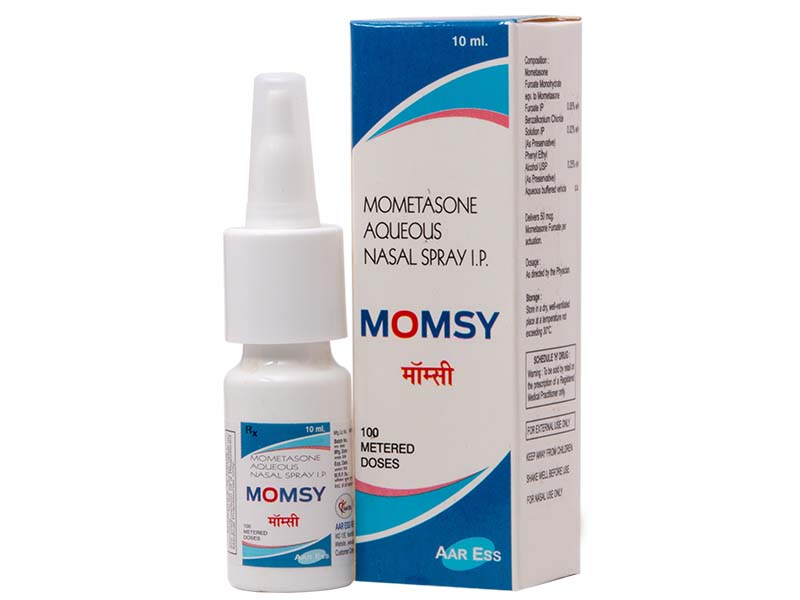
Mometasone furoate is a high-potency intranasal corticosteroid available for the treatment and/or prophylaxis of the nasal symptoms of seasonal allergic rhinitis (SAR) and perennial allergic rhinitis (PAR). In the EU, it is approved for use in patients aged > or =6 years and, in the US, it is approved as a treatment in patients aged > or =2 years and as prophylaxis in those > or =12 years of age.Extensive experience in both clinical trials and the clinical practice setting has firmly established the efficacy and good tolerability profile of intranasal mometasone furoate in children and adults with PAR or SAR. Thus, intranasal mometasone furoate is a useful first-line option for the treatment and prophylactic management of these conditions, including in children as young as 2 years of age in some countries and 6 years of age in others- Drugs. 2008; 68(12):1723-39.
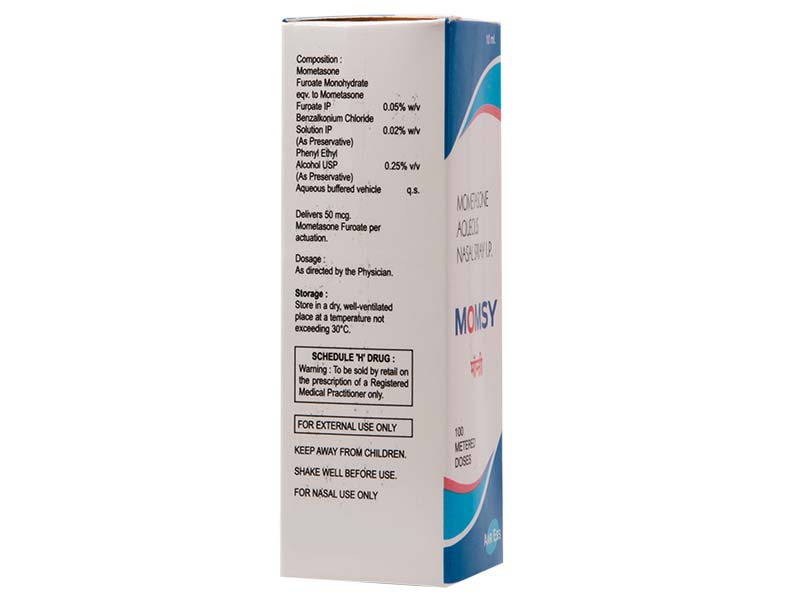
Mometasone Furoate Monohydrate
